 |
| http://www.ict4us.com/r.kuijt/images/en_oxidation_reduction.jpg |
The second type we learned of was synthesis. These reactions happens when two or more reactants come together and form one product. So A+B creates AB. Decomposition is the exact opposite of this, so there is one reactant breaking down to two or more products. This would then be AB creates A+B.
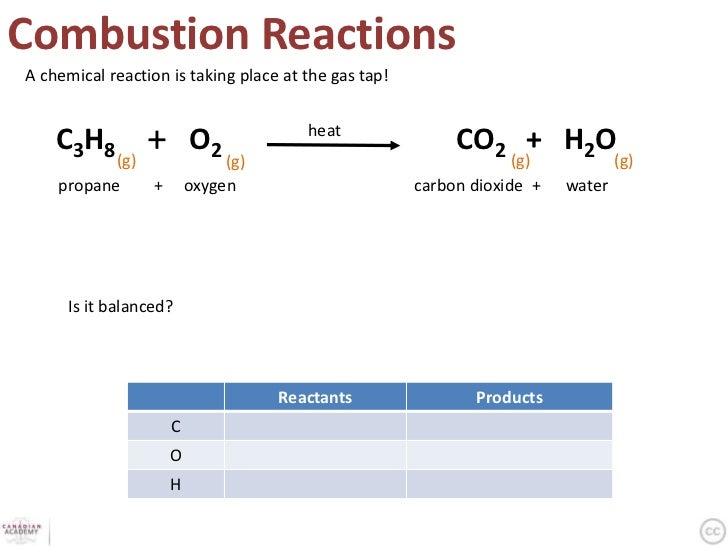
The final type we talked about was combustion reactions. In this type of reaction, when a hydrocarbon reacts with water, the products are always water and carbon dioxide. Here is an example:
Here are some video's to further explain these concepts:
https://www.youtube.com/watch?v=RX6rh-eeflM
https://www.khanacademy.org/science/chemistry/oxidation-reduction/redox-oxidation-reduction/v/oxidizing-and-reducing-agents-1
https://www.youtube.com/watch?v=RX6rh-eeflM
https://www.khanacademy.org/science/chemistry/oxidation-reduction/redox-oxidation-reduction/v/oxidizing-and-reducing-agents-1
No comments:
Post a Comment